Biomimetic Iron Complex Achieves TET Enzyme Reactivity
2021-06-28
David Schmidl, Niko S. W. Jonasson, Eva Korytiakov, Thomas Carell, Lena J. Daumann
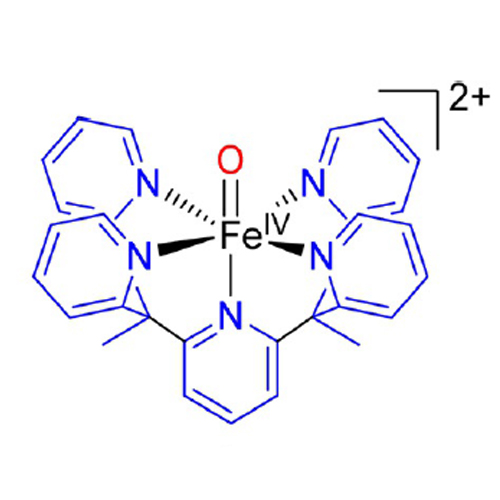
The epigenetic marker 5-methyl-2′-deoxycytidine (5mdC) is the most prevalent modification to DNA. It is removed inter alia via an active demethylation pathway: oxidation by Ten-Eleven Translocation 5-methyl cytosine dioxygenase (TET) and subsequent removal via base excision repair or direct demodification. Recently, we have shown that the synthetic iron(IV)-oxo complex [FeIV(O)(Py5Me2H)]2+ (1) can serve as a biomimetic model for TET by oxidizing the nucleobase 5-methyl cytosine (5mC) to its natural metabolites. In this work, we demonstrate that nucleosides and even short oligonucleotide strands can also serve as substrates, using a range of HPLC and MS techniques. We found that the 5-position of 5mC is oxidized preferably by 1, with side reactions occurring only at the strand ends of the used oligonucleotides. A detailed study of the reactivity of 1 towards nucleosides confirms our results; that oxidation of the anomeric center (1′) is the most common side reaction.